AML is one of the most common and deadly hematopoietic malignancies. The molecular and biological heterogeneity of AML poses significant challenges to the identification of novel agents to treat it and the optimal triage of patients for the most effective therapeutic regimens. Ex vivo models for drug sensitivity using cell lines and/or primary AML samples have been a fruitful resource for the exploration and discovery of novel compounds in the treatment of AML, and investigators have coupled genomic and transcriptomic profiling with ex vivo models in the hope to further enhance the reliability and accuracy of results. However, primary AML blasts are molecularly complex and fastidious, relying on extracellular matrix (ECM) and signals for optimal survival, making their use for large, high-throughput drug screens impractical. Available AML cell lines are more robust and less dependent upon the ECM but often harbor strong driver alterations (e.g., KMT2A rearrangements, TP53 mutations) that occur in only a small subpopulation of AML cases. Thus, current AML cell lines may not accurately predict agent sensitivity for most patients. Here, we report the results of a novel approach coupling cutting-edge proteomics with ex vivo drug sensitivity models to develop and describe a more diverse and representative set of patient-derived cell models for drug discovery and therapeutic positioning. Initial studies focused on proteomic biomarker signatures and cell lines to predict drug sensitivity for Venetoclax, which has become a stable component for treatment of older and unfit patients with AML.
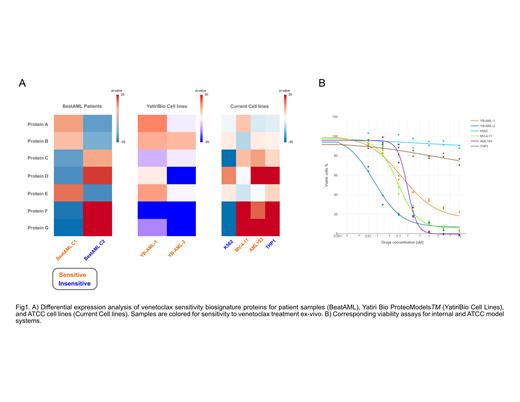
To identify a proteomic biosignature associated with Venetoclax sensitivity, we examined the Beat AML dataset of 210 patient proteomics coupled to ex vivo sensitivity assays. Beat AML patients clustered by unbiased proteomics which resulted in a cluster with a patient population significantly enriched in sensitivity to Venetoclax. A partial least squares discriminant analysis (PLSDA) was used to discover proteins driving the differentiation of Venetoclax sensitive and insensitive clusters. The resulting significant proteins describe a mechanism of altered DNA double stranded break repair and Interleukin-8 (IL-8)-CXCR2 signaling separating patients. These mechanisms were partially comprised of high-quality targets with differential analysis results shown in Fig. 1A. The identities of the targets will be revealed at the time of presentation as they are currently moving forward in preclinical studies.
To examine the potential predictive power of the Venetoclax sensitivity biosignature a set of novel ex vivo cell models were derived from a diverse independent population of patients obtained from FHCC/UW Myeloid Disease Repository. This population was not enriched for KMT2A or TP53 mutations. The proteomes of 90 diagnostic AML samples were examined and measured via a tandem mass tag protocol coupled with liquid chromatography with tandem mass spectrometry (TMT-LC-MS/MS). In addition, a proprietary set of novel ex vivo AML cell lines (n=17) representing the diverse genomic phenotype were generated from this same cohort of patients using proprietary media and pyrimidoindole derivative agonists of hematopoietic stem cell self-renewal. As with the primary samples, TMT-LC-MS/MS was utilized to quantify the proteome within the proprietary AML cell lines and 16 commercially available lines. Viability assays with Venetoclax were performed for all ex vivo cell models. Ex vivo cell models were then grouped based on the Venetoclax biosignature. The correlation between the proteomic biosignature for Venetoclax the measured sensitivity to Venetoclax was then examined for the ex vivo cell models (Fig. 1B). The results showed a strong correlation between the biosignature and actual ex vivo sensitivity to Venetoclax in proprietary cell models, but not in the 16 currently available cell models. Genetic drift of cell models from the primary AML patients was examined using differential proteomic analysis and no significant drift was found. The results indicated that the primary AML specimens harbored the Venetoclax sensitivity signature. Our findings reveal that the integration of unbiased patient proteomics with optimized ex vivo cell cultures can be used to identify sensitive patient subsets and exclude non-responders for novel therapeutics, as well as expanding patient access for currently approved therapies.
Disclosures
Jackson:Yatiri Bio: Current Employment, Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees. Stirewalt:Yatiri Bio: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees. Sorrentino:Yatiri Bio: Current Employment, Current equity holder in private company. Martinez de Bartolome Izquierdo:Yatiri Bio: Current Employment, Current holder of stock options in a privately-held company. Abbasian:Yatiri Bio: Current Employment, Current equity holder in private company. Bates:Yatiri Bio: Current Employment, Current equity holder in private company.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal